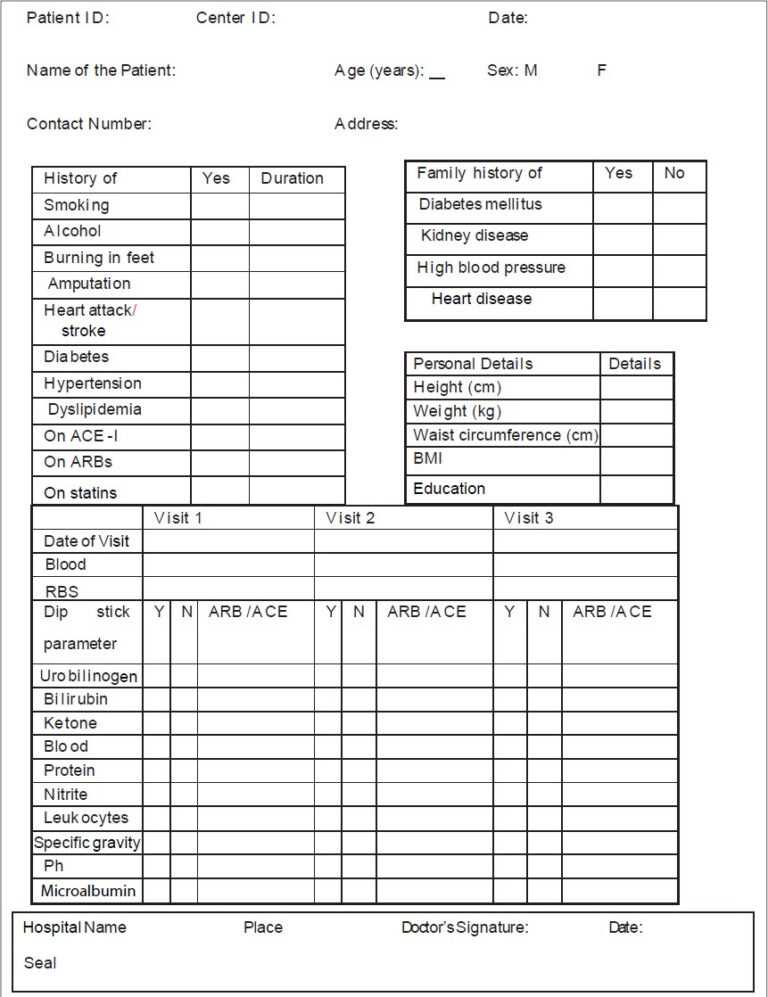
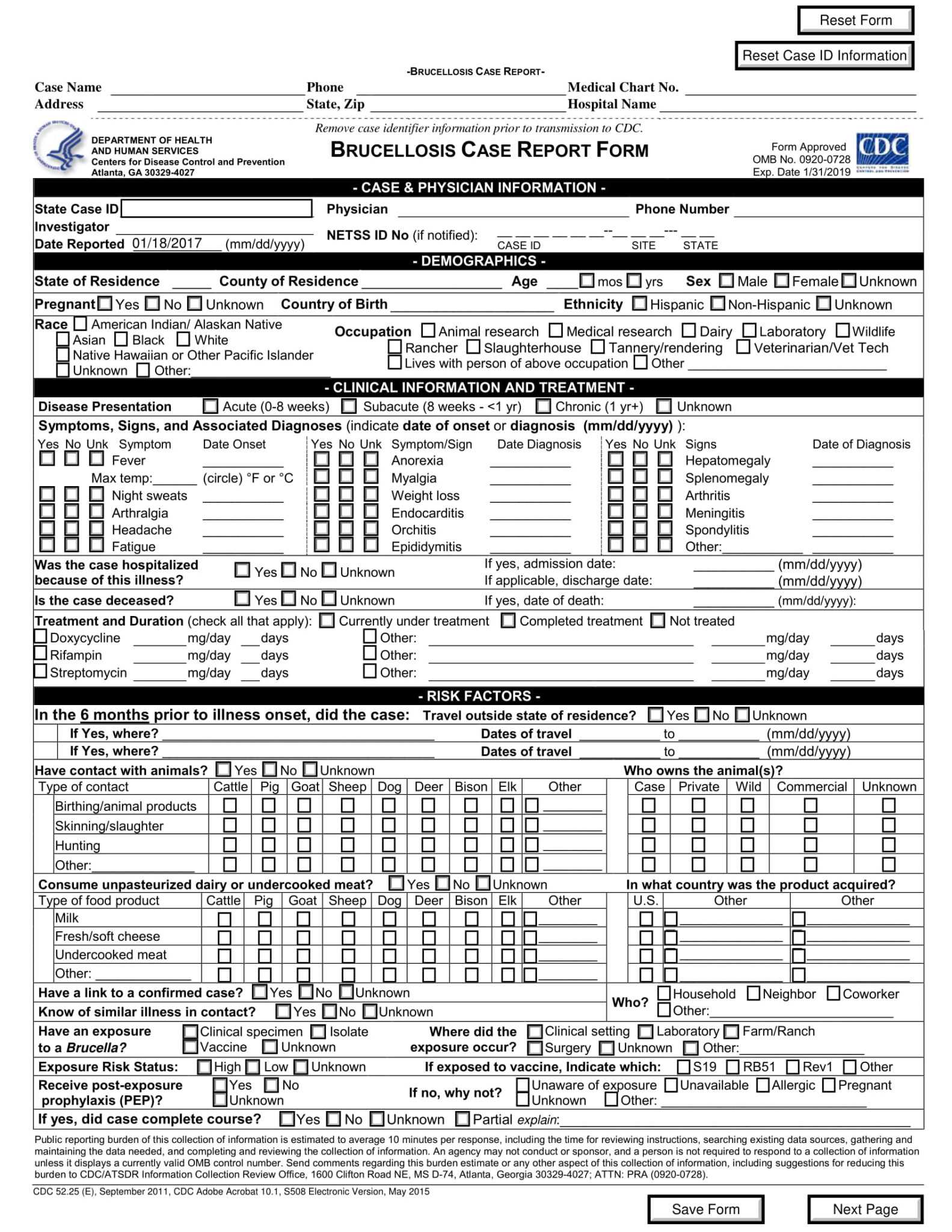
Case Report Form Clinical Trial
Case Report Form Clinical Trial - The case report form is the tool used by the. Web rdc case report form a controlled study of the ability of a traditional swedish smokeless tobacco product (“snus”) to. It supports healthcare professionals and. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. These templates are consistent with the fda cdash. Web gathering resources for case report forms is a great place to begin optimizing bottlenecks within your clinical trial operations. This article will discuss different case report. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also some common medical trial elements that are. Web august 15, 2014 when creating crfs in a clinical study build, it’s important to ask the right questions, but it is equally important to ask in the right way. Web this page provides links to commonly used clinical trial forms relevant to clinical trials.
This application is developed by clindox, a supplier of. Web rdc case report form a controlled study of the ability of a traditional swedish smokeless tobacco product (“snus”) to. These templates are consistent with the fda cdash. The case report form is the tool used by the. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also some common medical trial elements that are. Web this article is an attempt to describe the methods of crf designing in clinical research and discusses the challenges encountered in this process. Web crfweb is an electronic case report form (ecrf) for capturing data in pharmaceutical and medical device clinical trials. Web august 15, 2014 when creating crfs in a clinical study build, it’s important to ask the right questions, but it is equally important to ask in the right way. Web the primary purpose of this case report form template is to organize and track vital information for clinical trials efficiently. Web a case report form (crf) is an electronic or paper document which is used in a clinical trial to record the protocol and required information about each participant.
It supports healthcare professionals and. Web in a case report form, you can track the unique changes of each research subject as the clinical trial progresses. This article will discuss different case report. Web crfweb is an electronic case report form (ecrf) for capturing data in pharmaceutical and medical device clinical trials. Web this article is an attempt to describe the methods of crf designing in clinical research and discusses the challenges encountered in this process. This application is developed by clindox, a supplier of. Web a case report form (crf) is an electronic or paper document which is used in a clinical trial to record the protocol and required information about each participant. These templates are consistent with the fda cdash. Web gathering resources for case report forms is a great place to begin optimizing bottlenecks within your clinical trial operations. Web august 15, 2014 when creating crfs in a clinical study build, it’s important to ask the right questions, but it is equally important to ask in the right way.
Case Report Form Template Clinical Trials in 2021 Case study template
Web this page provides links to commonly used clinical trial forms relevant to clinical trials. Web electronic case report form initiative. Web gathering resources for case report forms is a great place to begin optimizing bottlenecks within your clinical trial operations. If you are looking to increase study start. Web agaclinicaltrials while each of our research trials at aga clinical.
Clinical Trial Report Template (2) TEMPLATES EXAMPLE
Web this article is an attempt to describe the methods of crf designing in clinical research and discusses the challenges encountered in this process. It supports healthcare professionals and. The case report form is the tool used by the. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web crfweb.
Case Report Form Template Clinical Trials
This article will discuss different case report. Web gathering resources for case report forms is a great place to begin optimizing bottlenecks within your clinical trial operations. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also some common medical trial elements that are. The case report form is the.
Case Report Form RIAT Support Center
This application is developed by clindox, a supplier of. Web the primary purpose of this case report form template is to organize and track vital information for clinical trials efficiently. The case report form is the tool used by the. Web a case report form (crf) is an electronic or paper document which is used in a clinical trial to.
What Is a Case Report Form? [ Importance, Tips, Samples ]
Web august 15, 2014 when creating crfs in a clinical study build, it’s important to ask the right questions, but it is equally important to ask in the right way. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web this article is an attempt to describe the methods of.
Journalbasics Of Case Report Form Designing In Clinical Inside
Web electronic case report form initiative. With this case report form, sponsors and medical. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also some common medical trial elements that are. This article will discuss different case report. Web the primary purpose of this case report form template is to.
The Basics Of Clinical Trial Centralized Monitoring with regard to
With this case report form, sponsors and medical. These templates are consistent with the fda cdash. Web this article is an attempt to describe the methods of crf designing in clinical research and discusses the challenges encountered in this process. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also.
FREE 15+ Case Report Forms in PDF MS Word
This article will discuss different case report. It supports healthcare professionals and. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web a case report form is a standardized questionnaire that is used during a clinical trial to collect data from each patient. With this case report form, sponsors and.
Free 15+ Case Report Forms In Pdf Ms Word For Case Report Form
Web in a case report form, you can track the unique changes of each research subject as the clinical trial progresses. Web a case report form (crf) is an electronic or paper document which is used in a clinical trial to record the protocol and required information about each participant. This article will discuss different case report. Web case report.
Free 15+ Case Report Forms In Pdf Ms Word pertaining to Case Report
Web the primary purpose of this case report form template is to organize and track vital information for clinical trials efficiently. These templates are consistent with the fda cdash. Web agaclinicaltrials while each of our research trials at aga clinical trials is unique in several ways, there are also some common medical trial elements that are. The case report form.
These Templates Are Consistent With The Fda Cdash.
Web rdc case report form a controlled study of the ability of a traditional swedish smokeless tobacco product (“snus”) to. Web gathering resources for case report forms is a great place to begin optimizing bottlenecks within your clinical trial operations. Web case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of. If you are looking to increase study start.
With This Case Report Form, Sponsors And Medical.
Web the primary purpose of this case report form template is to organize and track vital information for clinical trials efficiently. This article will discuss different case report. Web august 15, 2014 when creating crfs in a clinical study build, it’s important to ask the right questions, but it is equally important to ask in the right way. Web this page provides links to commonly used clinical trial forms relevant to clinical trials.
Web A Case Report Form (Or Crf) Is A Paper Or Electronic Questionnaire Specifically Used In Clinical Trial Research.
Web this article is an attempt to describe the methods of crf designing in clinical research and discusses the challenges encountered in this process. Web in a case report form, you can track the unique changes of each research subject as the clinical trial progresses. Web crfweb is an electronic case report form (ecrf) for capturing data in pharmaceutical and medical device clinical trials. It supports healthcare professionals and.
Web A Case Report Form (Crf) Is An Electronic Or Paper Document Which Is Used In A Clinical Trial To Record The Protocol And Required Information About Each Participant.
Web a case report form is a standardized questionnaire that is used during a clinical trial to collect data from each patient. Web electronic case report form initiative. The case report form is the tool used by the. This application is developed by clindox, a supplier of.


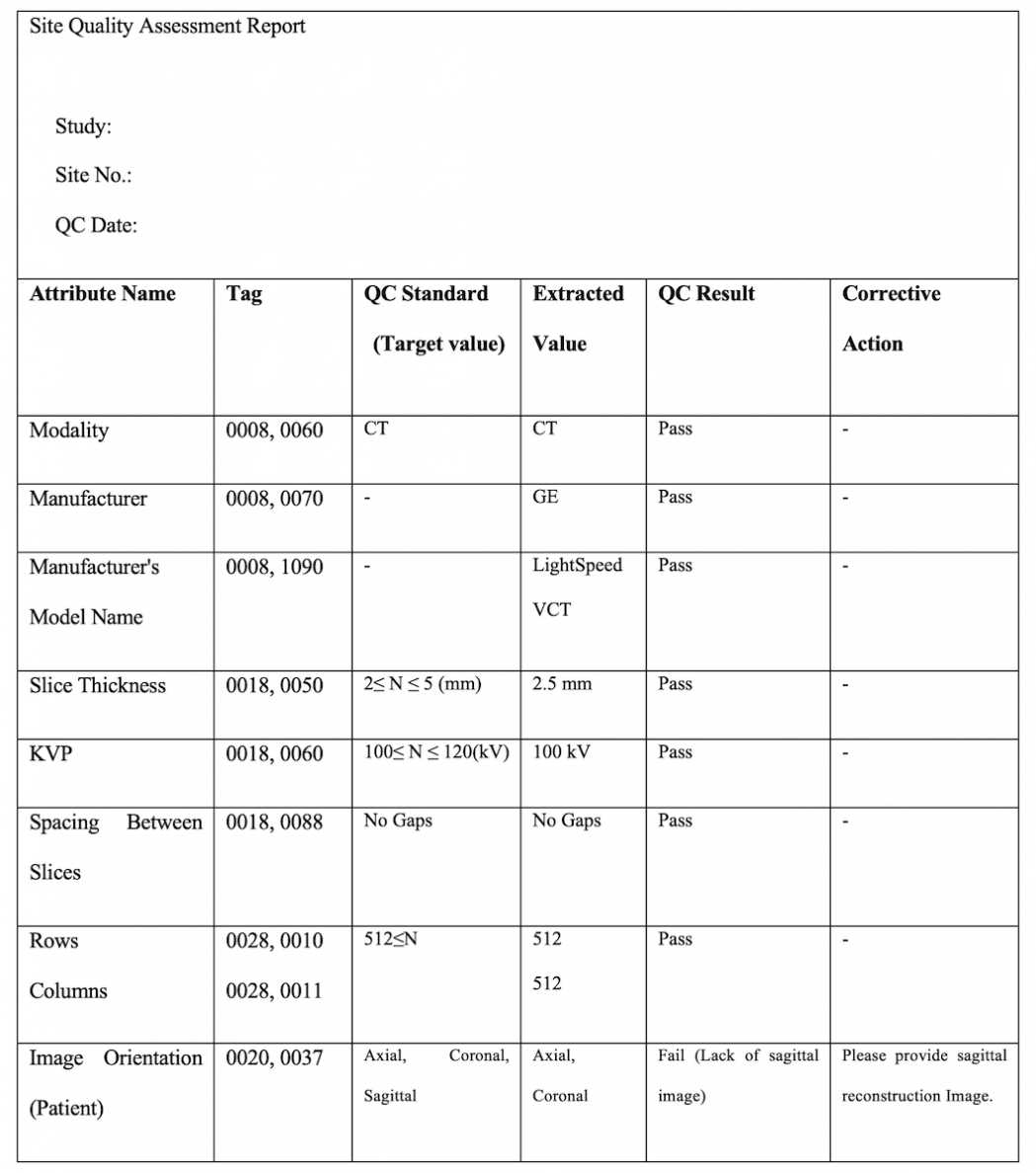

![What Is a Case Report Form? [ Importance, Tips, Samples ]](https://images.sampleforms.com/wp-content/uploads/2018/02/Confidential-Case-Report-Form-in-PDF-1.jpg)