How Many Covalent Bonds Can Hydrogen Form
How Many Covalent Bonds Can Hydrogen Form - Oxygen can form double covalent bond. This is the reason why h is always a terminal atom and never a central atom. How many covalent bonds do carbon nitrogen. Web a hydrogen atom with one electron and a chlorine atom with 17 electrons after bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is stable. Web 02/06/2017 chemistry high school answered a hydrogen atom has one electron. Terms in this set (28) one covalent bond a hydrogen atom has one electron. A solution with a ph of 7 is. A proposed explanation for a set of observations. Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; A hydrogen atom will typically form two covalent bonds because hydrogen has two electrons to.
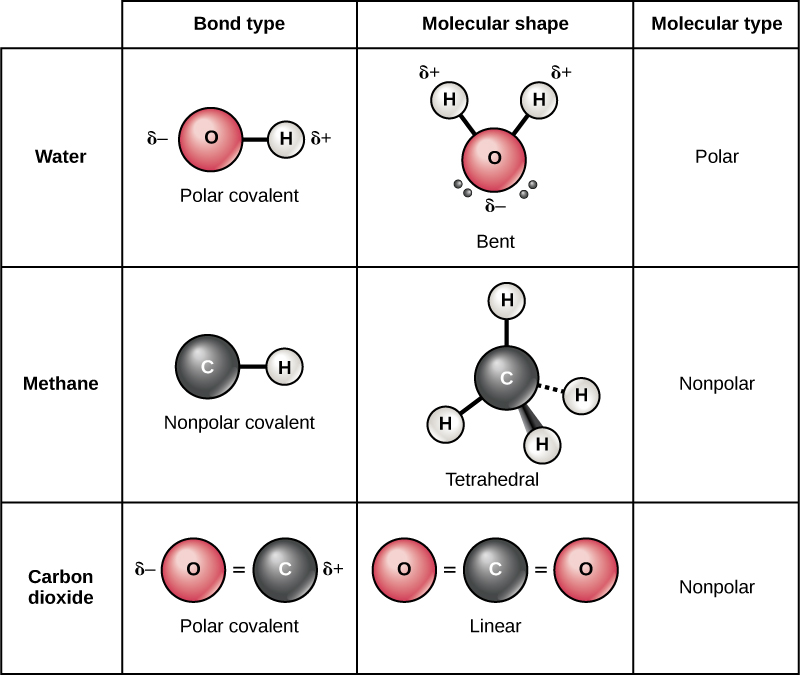
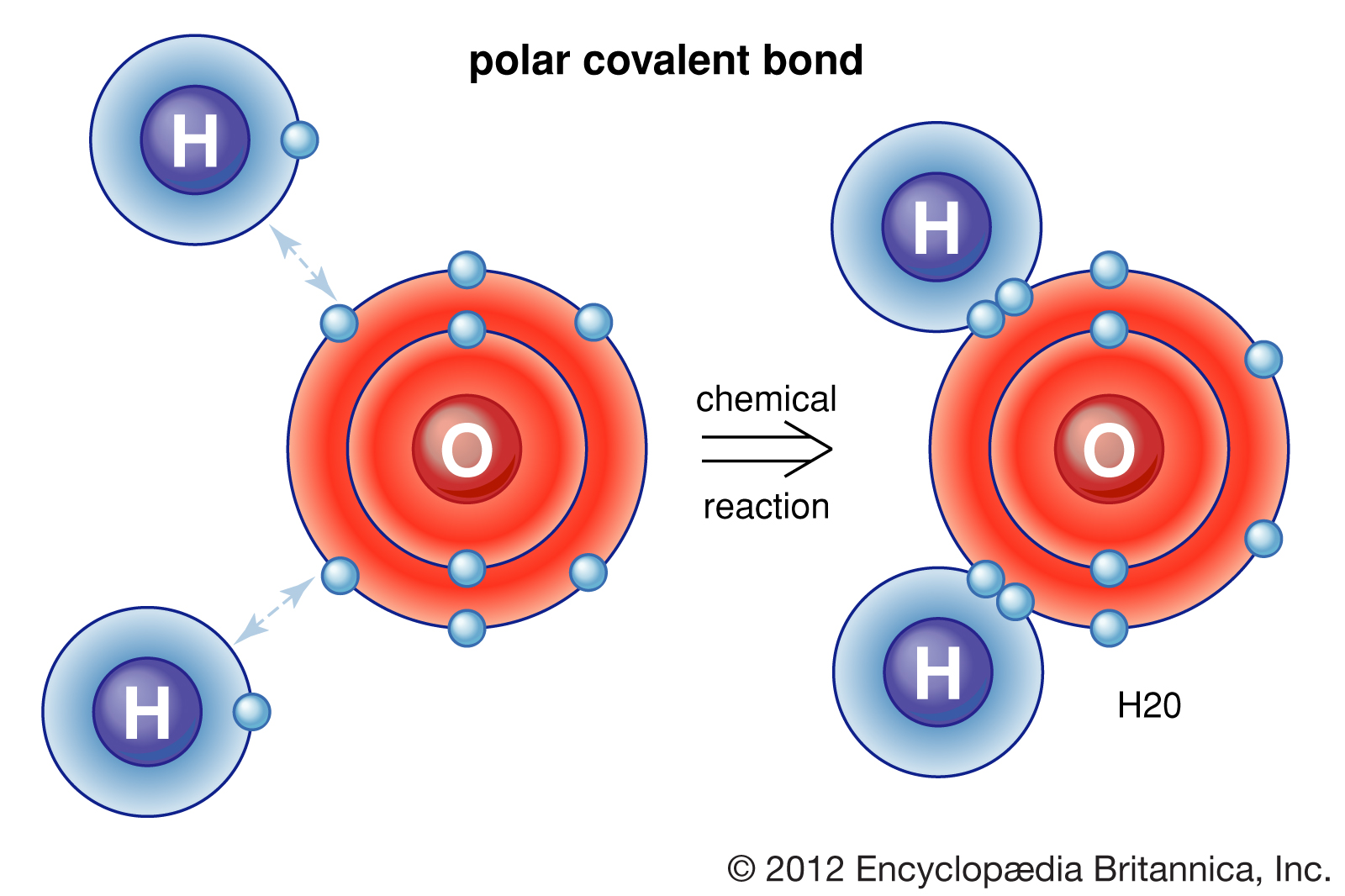
Oxygen can form double covalent bond. Hydrogen and helium are notable exceptions to the general rule that atoms share an appropriate. Web notice that each water molecule can potentially form four hydrogen bonds with surrounding water molecules: In the formation of a covalent hydrogen molecule, therefore, each hydrogen atom forms a single bond,. Web hydrogen only needs to form one bond. A proposed explanation for a set of observations. A solution with a ph of 7 is. A hydrogen atom will typically form two covalent bonds because hydrogen has two electrons to. Web 1 / 28 flashcards learn test match created by summer_32 project for last week of quarter 2. Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent.
Such a bond is weaker than an ionic bond or. Web a hydrogen atom will typically form two covalent bonds because hydrogen has two electrons to share in a bond. Web 1 / 28 flashcards learn test match created by summer_32 project for last week of quarter 2. A solution with a ph of 7 is. Terms in this set (28) one covalent bond a hydrogen atom has one electron. 1 shows the number of covalent. Web notice that each water molecule can potentially form four hydrogen bonds with surrounding water molecules: A proposed explanation for a set of observations. A hydrogen atom will typically form two covalent bonds because hydrogen has two electrons to. Web a covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms.
Is SiO2 Ionic or Covalent? Techiescientist
Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. How many covalent bonds do carbon nitrogen. Hydrogen can form only single covalent bond. Web 02/06/2017 chemistry high school answered a hydrogen atom has one electron. Web notice that each water molecule can potentially form four hydrogen bonds with surrounding.
Covalent Bonding (Biology) — Definition & Role Expii
Web 1 / 28 flashcards learn test match created by summer_32 project for last week of quarter 2. Hydrogen can form only single covalent bond. Web a covalent bond is formed between two atoms by sharing electrons. How many covalent bonds do carbon nitrogen. Oxygen can form double covalent bond.
2.2 Chemical Bonds Anatomy & Physiology
Nitrogen can form triple covalent bond. Hydrogen and helium are notable exceptions to the general rule that atoms share an appropriate. This is because the oxygen atom, in addition to forming bonds. Hydrogen can form only single covalent bond. This is the reason why h is always a terminal atom and never a central atom.
Bonds That Hold Water Molecules Together / Intermolecular Forces
Web a covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms. Web 1 / 28 flashcards learn test match created by summer_32 project for last week of quarter 2. This is because the oxygen atom, in addition to forming bonds. Covalent bonds can occur with most elements on the..
Covalent Bonds Biology for NonMajors I
Web hydrogen only needs to form one bond. For example, the hydrogen molecule, h 2, contains a covalent bond. In the formation of a covalent hydrogen molecule, therefore, each hydrogen atom forms a single bond,. How many covalent bonds can hydrogen form? Web hydrogen can form one covalent bond to have two valence electrons.
how many bonds does sulfur form
Hydrogen is an example of an extremely simple. Hydrogen can form only single covalent bond. How many covalent bonds do carbon nitrogen. This is because the oxygen atom, in addition to forming bonds. Such a bond is weaker than an ionic bond or.
How many covalent bonds can hydrogen, oxygen, nitrogen and carbon form
Hydrogen is shown in fig 2.28 with one electron. Web a covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms. Web how many covalent bonds can hydrogen form? Web formation of covalent bonds. Web 02/06/2017 chemistry high school answered a hydrogen atom has one electron.
Ch4 Polar Or Nonpolar Covalent Bond A CH4 B H2O C CF4 D CH3F Non
A solution with a ph of 7 is. Web how many covalent bonds will a hydrogen atom normally make? Web a covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms. Web a covalent bond is formed between two atoms by sharing electrons. Hydrogen is shown in fig 2.28 with.
[Best Answer] How many covalent bonds does carbon form if each of its
Web 02/06/2017 chemistry high school answered a hydrogen atom has one electron. Web a hydrogen atom with one electron and a chlorine atom with 17 electrons after bonding, the chlorine atom is now in contact with eight electrons in its outer shell, so it is stable. The number of bonds an element forms in a covalent compound is determined by.
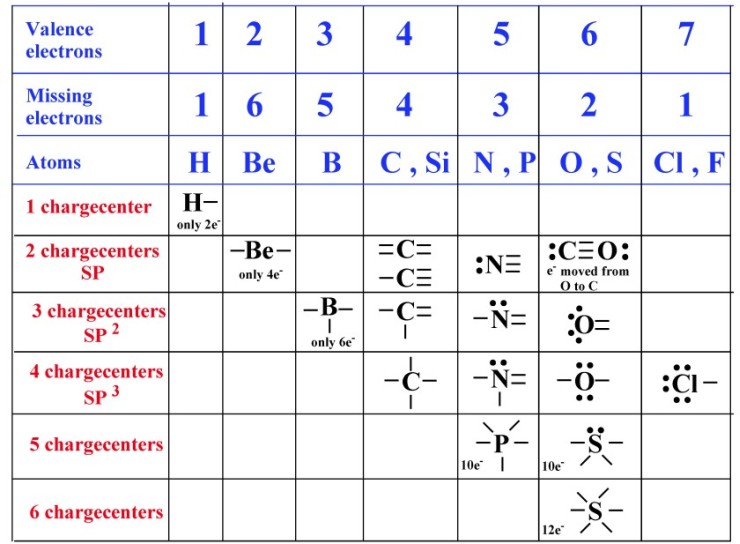
HONC 1234 ChemSimplified
How many covalent bonds do carbon nitrogen. A proposed explanation for a set of observations. Covalent bonds can occur with most elements on the. Hydrogen is shown in fig 2.28 with one electron. Web 1 / 28 flashcards learn test match created by summer_32 project for last week of quarter 2.
How Many Covalent Bonds Can Hydrogen Form?
Hydrogen is an example of an extremely simple. A solution with a ph of 7 is. Web a covalent bond is a chemical bond that comes from the sharing of one or more electron pairs between two atoms. Hydrogen can form only single covalent bond.
This Is Because The Oxygen Atom, In Addition To Forming Bonds.
Web formation of covalent bonds. This is the reason why h is always a terminal atom and never a central atom. Web how many covalent bonds can hydrogen form? A hydrogen atom will typically form two covalent bonds because hydrogen has two electrons to.
Web Hydrogen Can Form One Covalent Bond To Have Two Valence Electrons.
1 shows the number of covalent. In the formation of a covalent hydrogen molecule, therefore, each hydrogen atom forms a single bond,. Such a bond is weaker than an ionic bond or. The number of bonds an element forms in a covalent compound is determined by the number of electrons it.
Nitrogen Can Form Triple Covalent Bond.
Hydrogen is shown in fig 2.28 with one electron. Hydrogen and helium are notable exceptions to the general rule that atoms share an appropriate. Web 02/06/2017 chemistry high school answered a hydrogen atom has one electron. Both covalent and hydrogen bonds are forms of intermolecular forces.




![[Best Answer] How many covalent bonds does carbon form if each of its](https://us-static.z-dn.net/files/df7/e45f4693a936673d74acd687e2bb88cf.png)
