Xolair Patient Enrollment Form
Xolair Patient Enrollment Form - See full prescribing, safety, & boxed warning info. Web this service offers coverage support, patient assistance, and other useful information. Web download the forbearing consent form to begin enrollment with xolair access solutions. Blue cross and blue shield of texas. • adult and pediatric patients (6 years of age and above) with moderate to severe persistent asthma. Web patient enrollment forms | xolair access solutions forms and documents download the form you need to enroll in genentech access solutions. Genentech patient foundation provides free medicine to patients without. Web find xolair® (omalizumab) support for our practice, including financial supports, billing and distribution information, office support materials, & patient education resources. Web the first step is to have patients complete and submit the respiratory patient consent form. Web download of patient consent form to begin enrollment with xolair admittance choose.
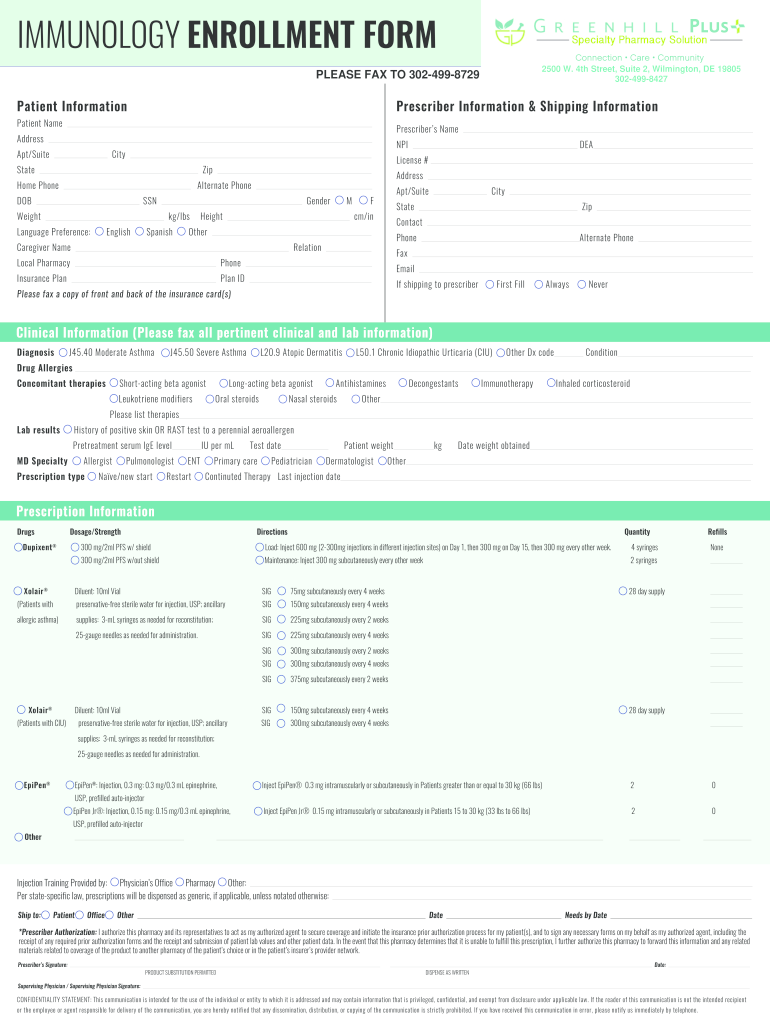
See full prescribing, safety, & boxed warning info. Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print). Web xhale+ program patient enrolment and consent form: Once completed, fax to the number indicated on the form. Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print). Web 1 of 2 prescription & enrollment form: In order to make appropriate medical necessity determinations,. Genentech patient foundation provides free medicine to patients without. Web download of patient consent form to begin enrollment with xolair admittance choose. Web patient enrollment and consent form xolair® (omalizumab) is indicated for:
Web with my patient solutions, you can: Web xhale+ program patient enrolment and consent form: Web this service offers coverage support, patient assistance, and other useful information. Web sign up to receive patient support resources, including information on getting started with xolair® (omalizumab). The bias introduced by allowing enrollment of patients previously exposed to. For patients prescribed prxolair® for moderate to severe allergic asthma (aa) or chronic idiopathic urticaria. Xolair ® (omalizumab) for subcutaneous use is an injectable prescription medicine used to treat: Please print and complete the forms below. Moderate to severe persistent asthma in people 6. Web xolair® (omalizumab) enrollment form xolair® (omalizumab) enrollment form fax completed form to:
Xhale+ Xolair Enrolment Consent Form Cloud Practice
Once completed, fax to the number indicated on the form. Ad proudly helping members navigate prescription assistance programs for 15 years! View benefits investigation (bi) reports; Web patient enrollment and consent form xolair® (omalizumab) is indicated for: Web the first step is to have patients complete and submit the respiratory patient consent form.
Enrollment Form For Xolair Enrollment Form
Ad proudly helping members navigate prescription assistance programs for 15 years! Your patient’s benefit plan requires prior authorization for certain medications. See full prescribing, safety, & boxed warning info. Xolair® (omalizumab) fax completed form to 866.531.1025. Ad visit the patient site to learn how the fasenra pen works.
Xolair Patient Consent Form 2023
Web xhale+ program patient enrolment and consent form: Review the dosing schedule and your administration options. Committed to helping patients access the xolair they have been prescribed. (1) documentation of positive clinical response to xolair therapy authorization will be issued for 12 months. • adult and pediatric patients (6 years of age and above) with moderate to severe persistent asthma.
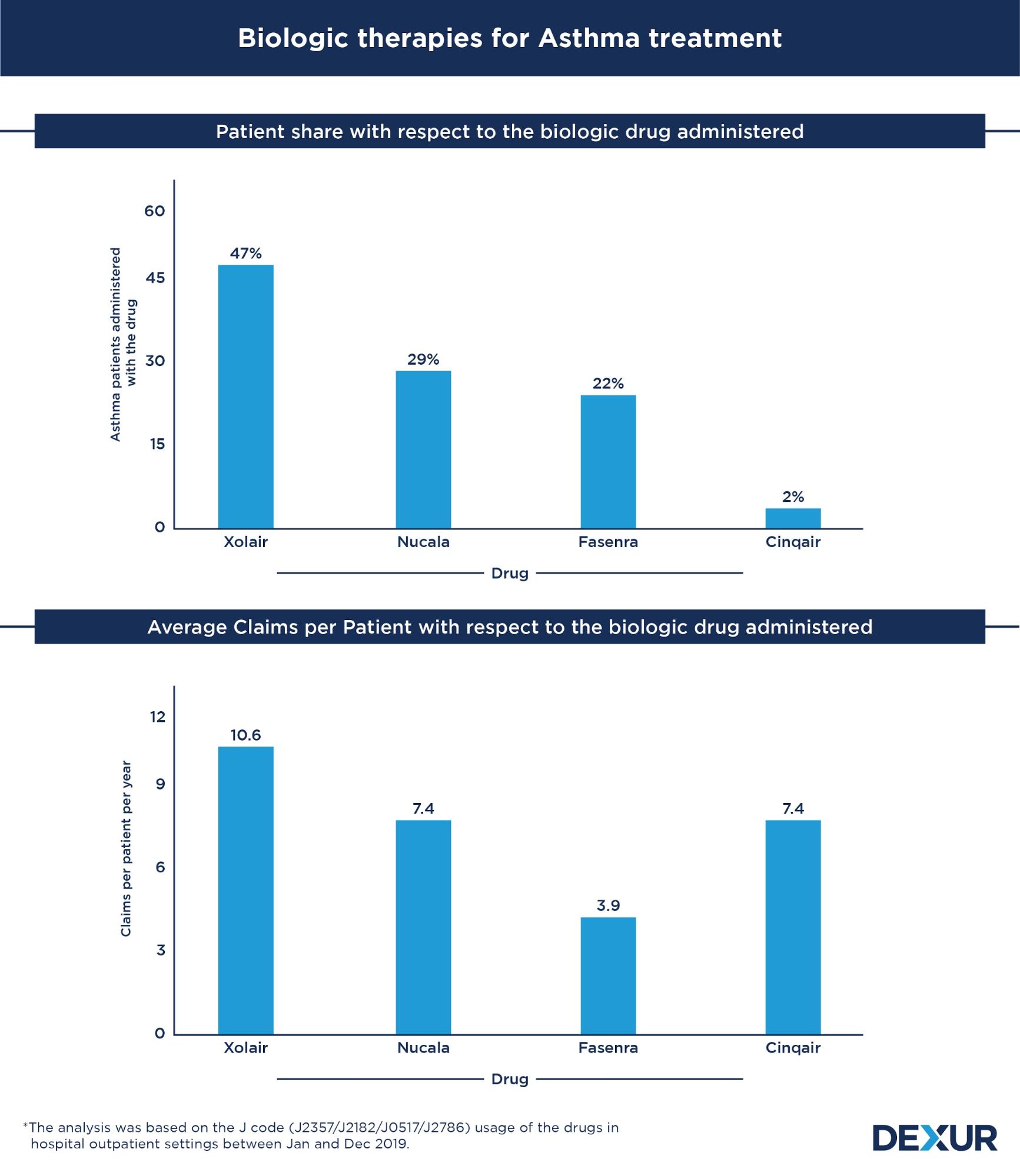
Xolair patient share was 1.6x more than Nucala and 2x compared to
Xolair ® (omalizumab) for subcutaneous use is an injectable prescription medicine used to treat: Web sign up to receive patient support resources, including information on getting started with xolair® (omalizumab). Web xolair will be approved based on the following criterion: View and track your patient cases; Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria.
XOLAIR Dosage & Rx Info Uses, Side Effects MPR
Web patient enrollment forms | xolair access solutions forms and documents download the form you need to enroll in genentech access solutions. The bias introduced by allowing enrollment of patients previously exposed to. Web with my patient solutions, you can: Web this service offers coverage support, patient assistance, and other useful information. Please print and complete the forms below.
Chronic Spontaneous Urticaria Treatment XOLAIR® (omalizumab)
Web 1 of 2 prescription & enrollment form: See full prescribing, safety, & boxed warning info. Web download of patient consent form to begin enrollment with xolair admittance choose. Xolair ® (omalizumab) for subcutaneous use is an injectable prescription medicine used to treat: Web the first step is to have patients complete and submit the respiratory patient consent form.
Xolair Enrollment Form Enrollment Form
Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print). Your patient’s benefit plan requires prior authorization for certain medications. Patient’s first name last name middle initial date of birth prescriber’s first. Genentech patient foundation provides free medicine to patients without. For patients prescribed prxolair® for.
Why Every Xolair Patient Should Keep an Allergy Journal IVX Health
Web with my patient solutions, you can: Xolair® (omalizumab) fax completed form to 866.531.1025. Web download the forbearing consent form to begin enrollment with xolair access solutions. Web download of patient consent form to begin enrollment with xolair admittance choose. Web find xolair® (omalizumab) support for our practice, including financial supports, billing and distribution information, office support materials, & patient.
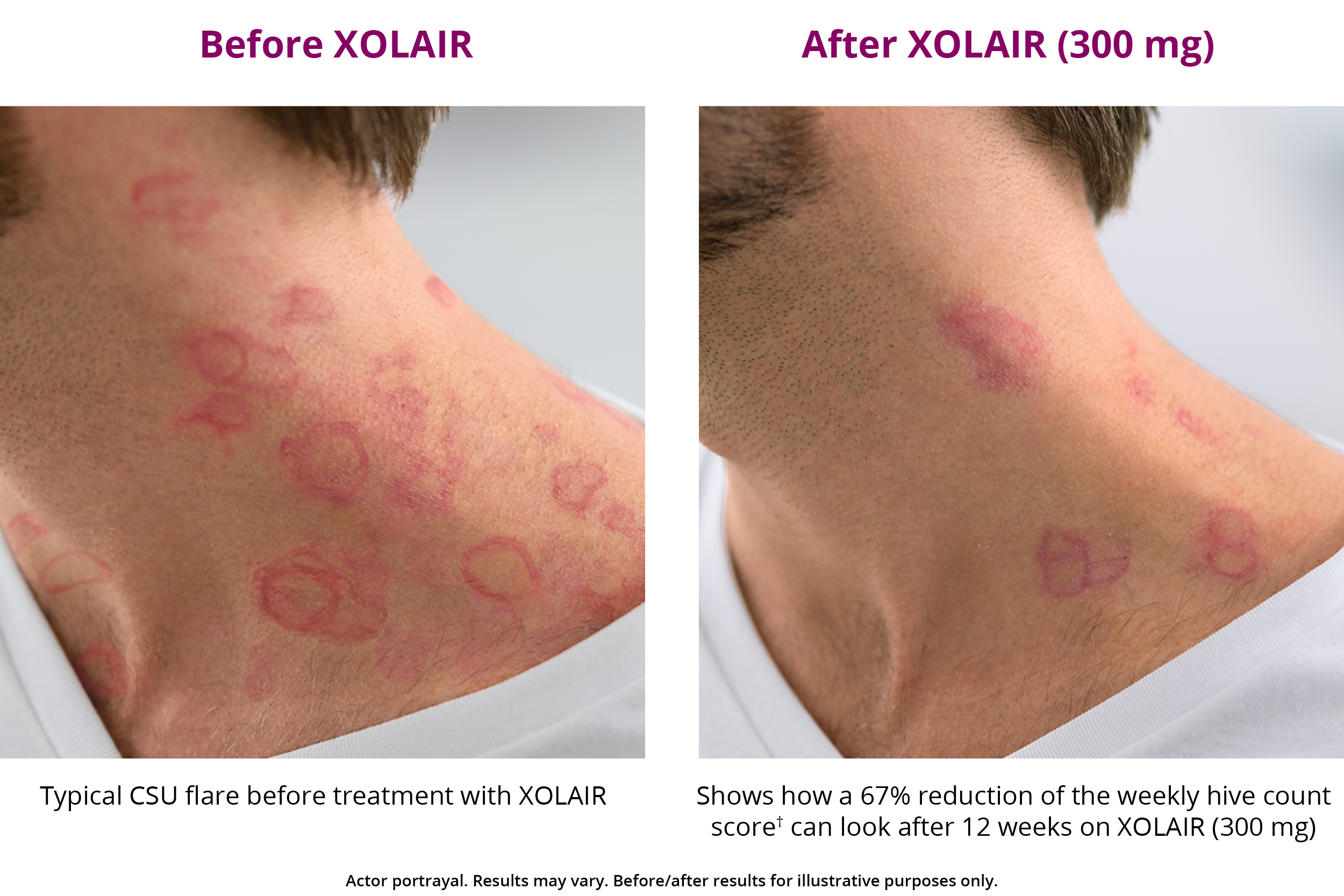
XOLAIR CSU Treatment Results XOLAIR® (omalizumab)
Genentech patient foundation provides free medicine to patients without. Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print). View and track your patient cases; Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled.
Xolair Dose Table Wallseat.co
Xolair ® (omalizumab) for subcutaneous use is an injectable prescription medicine used to treat: Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print). • adult and pediatric patients (6 years of age and above) with moderate to severe persistent asthma. Web xhale+ program patient enrolment.
Blue Cross And Blue Shield Of Texas.
Web this service offers coverage support, patient assistance, and other useful information. Web find xolair® (omalizumab) support for our practice, including financial supports, billing and distribution information, office support materials, & patient education resources. Web sign up to receive patient support resources, including information on getting started with xolair® (omalizumab). Web patient enrollment and consent form for patients prescribed prxolair® for chronic idiopathic urticaria (ciu), all sections must be completely filled out (please print).
Xolair® (Omalizumab) Fax Completed Form To 866.531.1025.
Web the first step is to have patients complete and submit the respiratory patient consent form. Ad visit the patient site to learn how the fasenra pen works. Committed to helping patients access the xolair they have been prescribed. Once completed, fax to the number indicated on the form.
Your Patient’s Benefit Plan Requires Prior Authorization For Certain Medications.
Ad proudly helping members navigate prescription assistance programs for 15 years! The bias introduced by allowing enrollment of patients previously exposed to. Xolair ® (omalizumab) for subcutaneous use is an injectable prescription medicine used to treat: Moderate to severe persistent asthma in people 6.
Patient’s First Name Last Name Middle Initial Date Of Birth Prescriber’s First.
Web xolair® (omalizumab) enrollment form xolair® (omalizumab) enrollment form fax completed form to: Genentech patient foundation provides free medicine to patients without. See full prescribing, safety, & boxed warning info. Web download of patient consent form to begin enrollment with xolair admittance choose.